Alactic & Aerobic (A+A) Training.
The A+A protocol uses the Alactic energy pathway of the cell to fuel the physical work of the repeat, or set. It then uses the Aerobic energy pathway to replenish the high-energy substrate of the alactic pathway, creatine phosphate, during the recovery period between repeats: Hence, “A” + “A”. Organizationally, the sessions of an A+A protocol consist of a variable number of repeats (NR), or work periods to be performed, each followed by an adequate rest and recovery period. The total volume of each session is the main variable. This protocol should only be used with high-power producing movements, and the kettlebell (KB) snatch lift one of the most effective.
In practice—what you do in the gym or your garage—is choose a high-powered movement and execute it with a purpose for 6-12 seconds, depending upon your current fitness. You then rest and recover for as long as is required so that you can again work at a high output. This can be as long as 2-3 min, or as little 1 min, again, depending on your fitness level. If you look at the diagram below, we are trying to employ the alactic system to fuel the work, and the aerobic system to fuel the restoration of the alactic system—it is during the recovery period that the aerobic system is challenged, and the overall duration of the session makes a great difference here.
The main variable to manipulate is the duration of each recurring session. An average of 30 repeats seems to work best, and you almost never want to actually complete 30—ebb and flowing under and over this average number will lead to better adaptation.
The protocol is blue-collar work through and through: punch in, do your work, and punch out. There are no “gym” goals; the only goal is to make you a stronger and more enduring you. It is work that is intense for short period of time, followed by a rest period while you prepare to do more work of the same kind. Conjure the images of loading a truck, chopping wood, farming, working on your own home improvements—you move to accelerate loads, then recover during your body weight movement back to the next load. The relatively short work period combined with a sufficient recovery period, in the context of extended sessions is what allows A+A training to increase strength-endurance while improving health. This method of training increases your biological resiliency rather than depleting it.
Repeats are strategically prescribed to allow a sufficient amount of recovery during the rest period so that you are able to go into the subsequent repeat performing at a high power level. If you believe the energy system science/philosophy and debates, repeats can target specific energy systems, causing them to adapt in positive ways, i.e., improved function. A+A repeats maintain the work period at a high output, and not continue too long such that output falls off. In biochemical speak, A+A avoids too much glycolysis, or, lactate-producing work.
Biochemistry.
All cellular fueling is accomplished by releasing the free energy contained within a molecule called, adenosine triphosphate (ATP). The ATP molecule is the energy currency of the cell: It is the money used to purchase energy for fueling work. Work does not just refer to muscle activity and movement; every type of cell in your body must accomplish physical work on a microscopic level, such as pumping ions across gradients.
The ATP molecule is a high-energy phosphate compound that can be chemically acted upon, producing different substrates while releasing energy. There is a small pool of ATP within each cell that it uses to fuel work. However, this pool is quite small and can only provide fuel for a short time. As your cell does work, the pool level drops but there are several systems in place that create ATP molecules from other substrates, restoring the pool level. The professional literature suggests that under no circumstances does the pool level drop below about 70% of its resting level. Fatigue sets in prior to this through many mechanisms, as running out of ATP would result in the death of the cell.
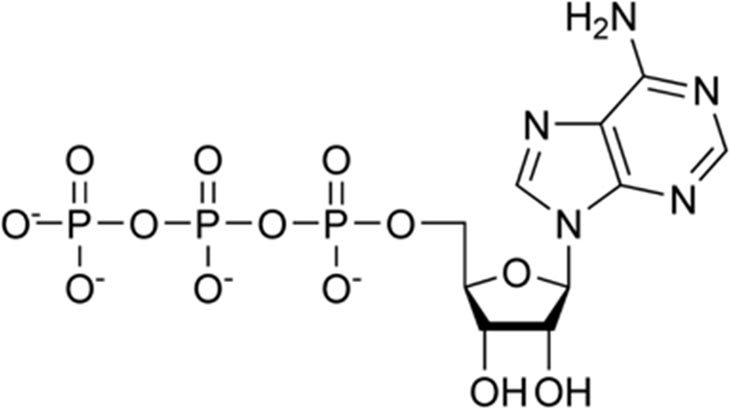
Above is a simple graphic of the ATP molecule. Without exploring too far into chemistry, you will note the three phosphate groups to the left. There is a lot of energy binding that last phosphate group to the package. What is known as cleaving in chemistry, this last phosphate group is removed from the molecule, releasing the energy stored in to the environment.
This splitting of the ATP molecule, called ATP hydrolysis, is the act of cleaving off this phosphate group. This can occur when ATP chemically reacts with a water molecule. The rate of this reaction occurring can be increased through the use of an enzyme: ATPase. Enzymes are proteins that can interact with atoms and molecules, speeding up the rate of their reactions but without changing the products of that reaction.
In this case, ATPase acts on ATP and a water molecule and converts them into:
- An adenosine diphosphate molecule (ADP)
- An inorganic phosphate compound (Pi)
- Hydrogen ions (H+)
- Heat
- And the goal: “Free energy”
Here is the reaction:
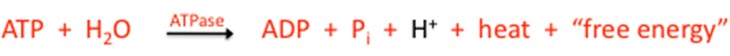
Note the hydrogen ion to the right of this chemical equation (in black). This is significant, as increasing the hydrogen ion content of a solution reduces its pH level, or makes it more acidic. This acidity—this increase in cellular acidosis—is likely the most dominant cause for a decrease in energy system output. The cell is placed in a state where it is unable to replenish ATP molecules at a rate that maintains a sufficient ATP pool level—one of the several ways that can lead to fatigue. Not unrelated, increased acid levels degrade proteins, the stuff you are made of. The three-dimensional shape of the protein molecule makes it very sensitive to hydrogen ions. On a systemic level, chronic acidosis might significantly contribute to ill-health and dysfunction of many varieties.
The raw materials needed to create new ATP are already present in the cell: the ADP and Pi molecules. Recall that these ingredients are formed during ATP hydrolysis, and the cell has systems in place that effectively recycle ADP and Pi back into ATP. Clever and efficient—energy is the missing ingredient in this biological stew.
The energy that is required to form new ATP molecules comes from the food we eat and/or (as in case of starvation) the physical structure of our bodies. The cell has three energy systems, or pathways, that we know of. These sets of chemical reactions take the products of digestion—the macronutrients in your food—and catabolize them, or break them down, harvesting the energy from within those molecules and using it to provide ATP with it’s “jolt” when rejoining ADP with Pi.
The energy pathways are known by several different names, depending upon the university you attend, the country you live in, or the author you are reading. A short list of these follows:
- The PCr energy system, also known as the alactic pathway, the phosphagen system, or the ATP-PCr system
- The glycolytic energy system, also known as the lactic pathway, glycolysis, or (erroneously) anaerobic glycolysis
- The oxidative pathway, also known as oxidative phosphorylation, the aerobic system, or mitochondrial respiration
The first two systems are classified as anaerobic, because they create ATP without the use of oxygen. The final pathway is defined as aerobic, because it requires oxygen to create ATP.
Alactic Energy.
The PCr energy system is not a “system” at all but a very simple chemical reaction that acts on a phosphocreatine molecule (hence the name of the pathway). Note the following reaction:
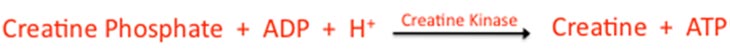
The creatine phosphate molecule has a high-energy phosphate component it. This phosphate is cleaved from the molecule, and then combined with ADP and a hydrogen ion. If you have been paying attention, you will note that this process actually alkalizes the cell (increases its pH) because it removes hydrogen from the environment. But not to such a degree that it is worth anything but a mention here. However, for single-celled creatures, this strategy could be very important!
Like the ATP molecules that comprise the ATP pool, creatine phosphate is a substrate present in the cell, but in very short supply. Remember that an alternative name for this pathway is ATP-PCr. This title is simply lumping the ATP pool together with the above reaction. This first pathway of generating ATP is quick and simple—remove energy from phosphocreatine and combine it with ADP and H+.
This is where the traditional textbooks stop. There are two additional cellular reactions that Baker, McCormick, and Robergs include as part of the PCr system:
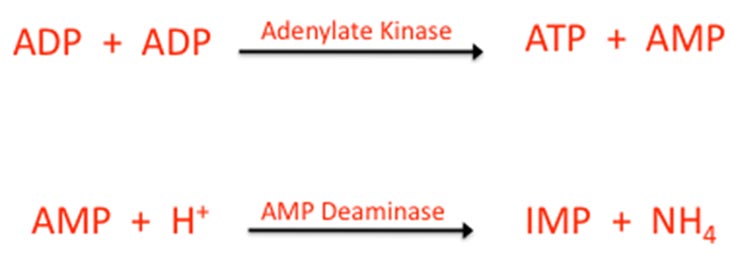
Dr. Keyser tells us that increasing ADP levels and decreasing ATP levels in the cell lead to a decreasing ATP:ADP ratio; and this can also lead to fatigue. Increasing ADP level causes adenylate kinase to go to work on ADP, reforming ATP and creating a molecule called, adenosine monophosphate (AMP). This can be seen in the first reaction.
In the second reaction, AMP combines with hydrogen to form inosine monophosphate (IMP) and ammonia (NH4), which the cell, and body, must get rid of.
The point in introducing these additional reactions is:
- AMP activates some of the enzymes in glycolysis (the next pathway we will discuss)
- These three reactions together may produce enough ATP to power both resting level activity, and supra-resting level activity, possibly changing how we currently think about bioenergetics
- These two additional reactions suggest that PCr system function might also keep ADP, AMP, and H+ levels down, helping to prevent fatigue
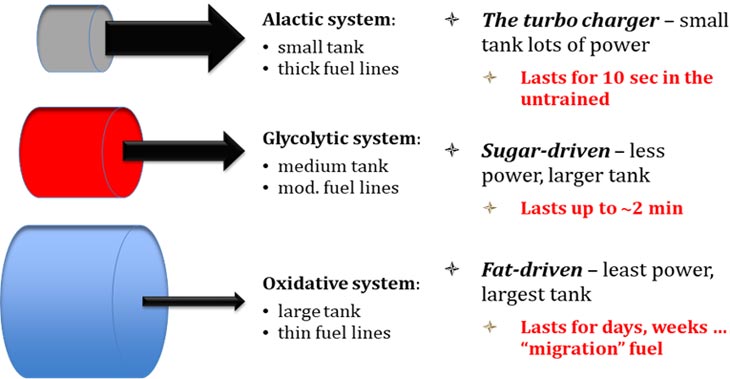
This system is very powerful, but with little capacity. It can restore ATP quickly, but only in small amounts. It is thought by exercise physiologists that this system is exhausted after about 10-15 seconds, but can power the most intense of muscular contractions, like sprinting and heavy weightlifting. It is important because it is used in most sport applications: Intermittent and short high-intensity bouts of activity repeated after longer intervals of active or passive rest, describes many of the games that we play. Think about what an individual actually does in basketball, American football, soccer, ice hockey, etc. A lot of jogging/walking separated by very short but very intense sprints/movements… this energy system plays a very important role in short and intense activity, and its ability to recover is central to success in sports.
Glycolytic Energy
The next energy pathway is glycolysis, which literally means splitting of sugar. This lactate pathway acts only on sugar molecules (glucose or glycogen)—so, we are now using the energy from the food we eat to fuel cellular function. As compared to the three, single-function chemical reactions of the PCr pathway, glycolysis is a much more complicated series of reactions, and the chemical reactions of the lactate system work on glucose in a linear manner.
Glycolysis removes the energy from either the glucose or the glycogen molecule (the storage from of glucose) and uses it to form ATP. The main molecular product of these reactions is pyruvate; or lactate, depending upon which scientist’s idea that you purchase. There are two phases to glycolysis: The preparatory phase, and the pay-off phase. To keep things neat and easy to understand, I have included only the molecules being acted upon (denoted by the blue arrows), and the required/resulting substrates of several individual reactions (denoted by the black arrows) in the figure below. The preparatory phase creates two molecules of glyceraldehyde 3-phosphate that can be used in the pay-off phase. In this first phase, ATP is required for this conversion—two, if we begin with a glucose molecule, and one, if we begin with a glycogen molecule. Read that again. You can see this on the top left of the figure.
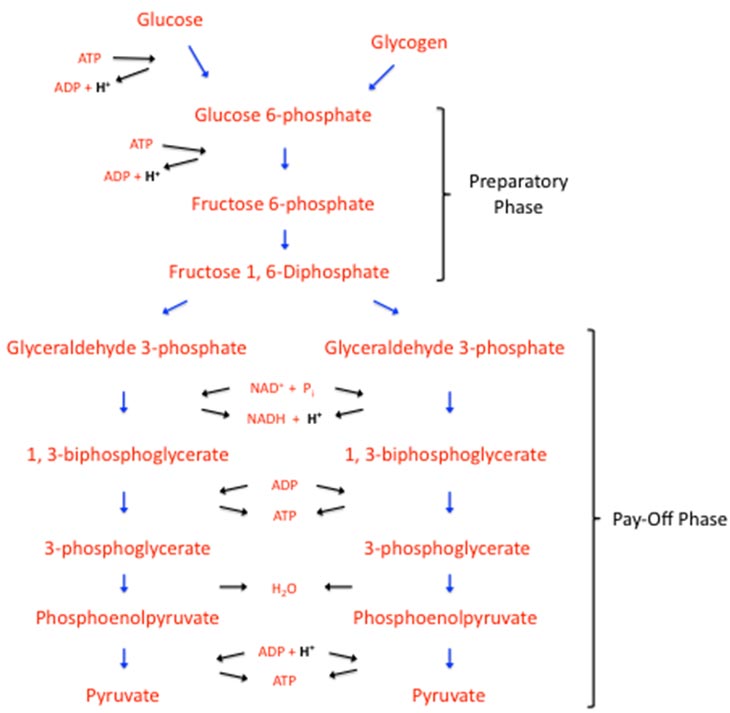
In the preparatory phase, ATP hydrolysis adds ADP and hydrogens to the environment, twice as much if using glucose, which may lend to the idea of not completely foregoing starchy carbohydrates. It is the pay-off phase of glycolysis that creates ATP. Through several additional chemical reactions, each of those two glyceraldehyde 3-phosphate molecules is converted into one molecule of pyruvate. You will note this in the figure down the middle of the pay-off phase with the corresponding reactions.
A positively charged molecule, nicotinamide adenine dinucleotide (NAD+) and an inorganic phosphate molecule is needed to convert glyceraldehyde 3-phosphate to 3-biphosphoglycerate. As a result of this reaction, NADH (the reduced form of NAD+) and a hydrogen ion are formed. The reason this is pointed out will become clearer later. In the next reaction, an ADP is used and we get our first ATP. The next reaction down produces water; and the final reaction uses ADP and H+, and produces our other ATP molecule.
The total yield for glycolysis is:
- 3 x ATP
- 2 x H+
- 2 x pyruvates
- 2 x NADH
- 2 x water molecules
…unless you begin with glucose, then the result is “2 x ATP and 1 x H+”, and everything else is the same. So, you not only create less ATP through glycolysis when using a glucose molecule, but you also introduce additional acid to the cell.
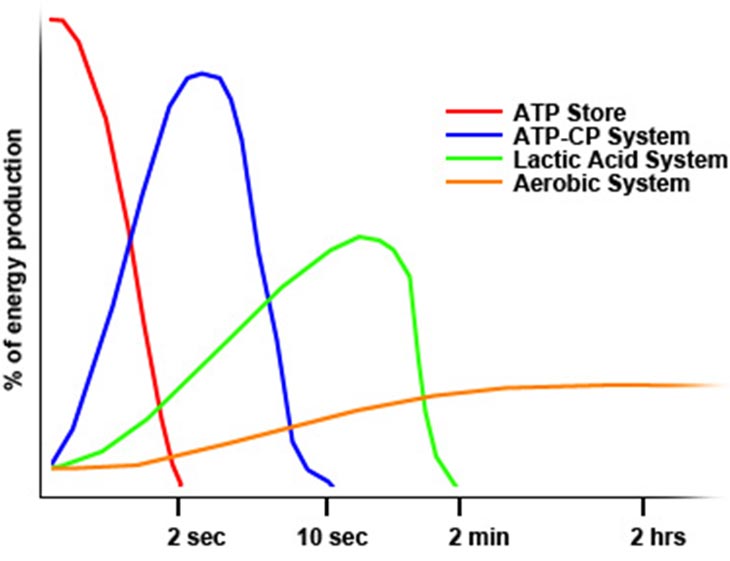
The glycolytic pathway has less power than the PCr pathway, but it has more capacity. That is, it can’t peak as high in production of ATP, but it can do so for a longer period of time. You can see in the figure below how glycolysis (the lactic acid system) burns out around the two-minute mark when engaged in all out activity from rest.
Being a very ancient energy pathway, the one that allowed animal life to begin, glycolysis is actually a very inefficient way to produce energy—only 3% of the available energy is extracted from the sugar molecule. But, nature has adapted to this problem…
The Mitochondrion: Oxidative Energy
The mitochondrion is thought to once be a separate organism from the cell and the entire reason that multi-cellular animal life is so prolific today. In a powerful example of biological symbiosis, the mitochondrion feasts on pyruvate, lactate, fatty acids and hydrogen electrons, and produces CO2, water, and ATP. The mitochondrion has its own structure, membrane and DNA. It can replicate itself in a process similar to asexual reproduction, divorced from the activity of the eukaryotic cell within which it resides.
The medical literature has been revealing the association between the health of this “organism” and that of the host organism—you! You might be reading this article for a different reason, but you should be extremely interested in the health of your mitochondria.
Inside the mitochondrion is where the magic happens: Oxidative phosphorylation. This is a very complex system of ATP restoration that we likely do not fully understand. It occurs in two phases: The Citric Acid Cycle, and the Electron Transport Chain. Also known as the Kreb’s cycle, the citric acid cycle is a series of eight reactions that is spun through twice. The feeder molecule for the citric acid cycle is called acetyl CoA and it is the only molecule that this system can work on, so whatever the input—fatty acids, lacate, pyruvate, ketones—they all must be converted to acetyl CoA first. It’s simplistic to look at the citric acid cycle as a preparatory phase for the electron transport chain (ETC), but it fits with the observations. What the citric acid cycle does is harvest hydrogen protons and electrons, and the associated energy, for use in the ETC, where ATP is formed.
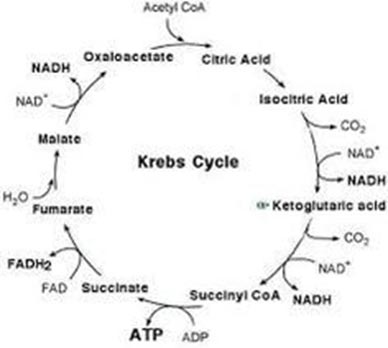
You can see in the above figure that NAD+ is required to carry the hydrogens to the ETC. Recall this function from glycolysis: NAD+ are the carrier molecules for hydrogens, and without enough NAD+, none of these systems can function. NAD+ might be the one very important substrate that few exercise scientists are talking about—it is effectively the bottleneck of the energy systems. When oxidative phosphorylation backs up, anaerobic metabolism must make up the difference, or movement intensity must drop off. The “fix” to maintain anaerobic respiration—which means to free up more NAD+—is the formation of lactate.
The ETC is where the pay off is. Every resource that I have read discussing the ETC uses terms like, “seems to”, “this suggests”, and “this or that may occur”, so let me state now that it is likely we have little idea what is actually happening in there. That said it seems that within the mitochondrion, there are two compartments separated by a membrane. Along the membrane are a series of structures, called cytochromes, and pumps, similar to the ion pumps of the cell membrane. The electrons delivered to the ETC by NADH move down the cytochromes in a train-like fashion, one to the next with a beginning and an end. Each relocation of an electron transfers some energy. Some of this energy is used to pump protons, also delivered by NADH, across the inner membrane, setting up a gradient. This gradient results in stored electrochemical energy, a form of potential energy. This energy can then be used to synthesize ATP molecules, by phosphorylating ADP, or adding an inorganic phosphate to an ADP molecule.
You might be wondering how oxygen fits into this schema… well, when electrons are added to a molecule or atom, it is called “reduction” and when they are removed from one, it is called “oxidation”. The cytochromes of the ETC act like atoms or molecules in this case, and function similar to a bucket brigade. Which looks like this:

Just like the firemen of the past in this picture, the cyctohromes hand the electrons to each other, so, a cytochrome has to hand his electron off before he can grab another. This oxidation-reduction reaction series continues on until the last cytochrome has his bucket of water, I mean, electron. This cytochrome would remain in this reduced state if there was no fire to dump his bucket on—this is the function that oxygen serves. O2 acts as the last man on the bucket brigade line, accepting the electron from the final cytochrome so that aerobic respiration can continue. Coupled with the protons that are diffusing back across the inner membrane in the phosphorylation process, the hydrogen atom is reformed, but in the water molecule: H2O.
It is ingrained in our language, at this point, to say that aerobic metabolism is limited by the availability of O2. That aerobic means, “with oxygen”, does not mean it is the governor of this system. It is probably not a lack of O2 (however much it may contribute) that causes you to outwork your aerobic system and move to using primarily the anaerobic system for output. Again, it is far more likely that it is a lack of NAD+ to carry hydrogens where they are needed. Unlike the media would have you believe, O2 is plentiful in our atmosphere, lungs, blood and tissues (in otherwise healthy individuals).
In summary, all substrates and foodstuffs are converted to acetyl CoA within the mitochondrion. Acetyl CoA is then run twice through the citric acid cycle in the mitochondrial matrix, extracting hydrogen protons and electrons for the electron transport chain. The ETC is a bucket brigade that collects the potential energy from the hydrogens and uses it to create ATP. The hydrogens are then passed on to oxygen molecules and form water. The oxidative system results in a rather “clean burn”: ATP, carbon dioxide, water, and heat. This is the beauty and magic of nature: far before we were able to look and see the mechanics of mitochondrial energy production, we built dams across rivers and used the potential energy of the weight of moving water to crank turbines which then created electrical energy. In very much the same way, the mitochondrion uses the potential energy of the weight of moving protons to crank a turbine and create ATP. In fact, this turbine is the smallest known “machine”.
The ATP yield from this process is 32-38 molecules depending upon what textbook you refer to. This grants you 34% of the energy from the original glucose molecule, as compared to the 3% extract from glycolysis. The remaining 66% of the energy in sugar is transferred as heat.
The “Lactate System”.
The quotes are up there because I don’t believe that generating lactate from pyruvate is conventionally understood as a system. Remember the two pyruvate molecules as a result of glycolysis? If there is too much acid hanging around, the pyruvate molecule can accept two hydrogens and convert to a lactate molecule. In the figure below, we see that one of the hydrogens is received from a NADH molecule, reducing it back to a NAD+ molecule. This is thought to allow glycolysis to continue… remember that NAD+ is required to receive a hydrogen ion as part of the first reaction of the pay-off phase.
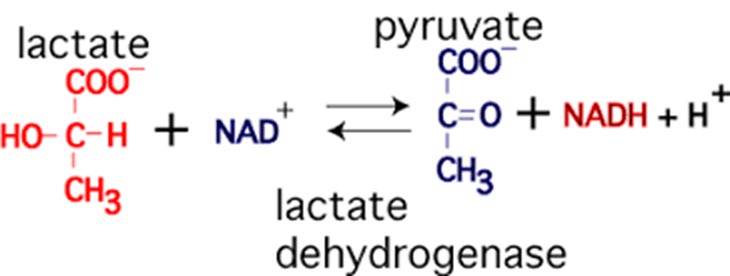
Forming lactate functionally reduces the pH concentration of the cell, is a mechanism for glycolysis to continue to function, and creates a molecule that can be shuttled out of the cell for use elsewhere. Moreover, lactate can also be shuttled into the mitochondrion to be converted to acetyl-CoA and used to produce ATP. Is lactate production a buffering strategy, or is it meant to allow glycolysis to continue? Probably both.
Lower blood lactate levels during exercise is associated with anti-glycolytic training and less of a biologic cost; higher blood lactate concentrations is associated with the opposite effect: Higher biological cost and increased stress levels.
Energy System(s) Interaction: Muscle fiber types.
It is difficult to have a discussion about energy system interaction without discussing muscle fiber types—so let’s begin there. From a practical standpoint, this is all that matters here.
Muscle tissue consists of many types of cells, or fibers. These fibers are known as fast twitch—those that can produce a lot of force; and, slow twitch—those that cannot produce a lot of force. Although science has mapped at least three different twitched fibers, if you are familiar with biology, it is far more likely that there are many speeds of fibers that exist on a continuum, from slowest to fastest. (Note that to my knowledge, this last statement is not supported by research). This continuum might look like this:
Slowest >>> Slower >>> Slow >>> Fast >>> Faster >>> Fastest
The slower fibers are more aerobic, meaning that to a major extent, they are structured to preferentially produce force with the use of oxygen. The important qualities of an aerobic muscle fiber are:
- It depends upon oxygen to properly function
- It is not capable of producing a lot of force
- It has a lot of stamina
The less aerobic a muscle fiber is, the more it depends on sugar to create force. These faster acting, anaerobic muscle fibers have the following important qualities:
- They require glucose (or glycogen) for function
- They can produce a lot of force
- They do not have a lot of stamina
Muscle fibers are recruited in a particular way: From the slowest (weakest) to the fastest (strongest), based on the needs of the action. So, easier movements recruit the slowest of fibers, and most forceful movements will recruit all the fibers on the continuum. I.e., walking will recruit the slowest of fibers: Sprinting, the fastest (as well as the slower ones). The point is:
The main driver that determines whether an activity is aerobic or anaerobic is fiber type recruitment.
I think that this point is lost in common energy system discussion. An activity is more aerobic or anaerobic based on the fiber types that are available at the time of movement. Consider the vast system of outputs in this respect:
- If you begin mall-walking, the slowest fibers are recruited and the activity is mostly an aerobic one; however, to actually get your started, there is an anaerobic “boost”
- If you pick up the pace to a semi-brisk walk, there may or may not be a boost of anaerobic metabolism based on the strength and endurance of your slower muscle fibers (in the extremely detrained or untrained, this already can be a mostly anaerobic activity)
- If you push the accelerator to the floor of your walking gait, chances are your slower fibers and so the aerobic pathway can cover this action
- If you break into a slow jog, the sharp increase in total force production required will no doubt enlist faster fibers, increasing anaerobic pathway involvement
- Depending upon your training state and the duration of movement at this point, your slower fibers might be able to cover down on your jog
- If you continued to jog at this slow speed, when your slower fibers powering this activity fatigue, faster fibers will cover down, causing the activity to be more and more anaerobic as you move forward (this can be seen on a heart rate monitor, as it begins to grow higher and higher)
- If, however, your slower fibers can endure, you will remain mostly aerobic
- If you hit hills or turn into the wind, and want to maintain the same speed, faster fibers will be recruited, temporarily increasing anaerobic metabolism
- If you speed up, faster fibers again must be recruited to cover the additional force production cost
- At any point that reduce the force production required, i.e., you slow down; and your slower fibers are not fatigued, you will be more and more aerobic
- And this will go on and on…
Let’s look at this from the perspective of high force production from the onset, say the 5-minute snatch test with little-to-no warm-up:
- The faster fibers (but probably not the fastest) will be recruited, making this a wholly anaerobic event
- As these faster fibers fatigue, the fastest fibers will begin to be recruited
- As these fibers fatigue, the only fibers left are slower; making the event more and more aerobic as time goes on
- If these slower fibers are not very strong, you may have to stop the event all together; if they are, however, your pace and intensity will simply have to decrease
- If you pace this properly, the fastest (or fast”er”) fibers might be able to recover enough during the activity, which allows for a sprint to the finish!

Recall the chart above… it is a pretty good representation of the how each energy pathway works in isolation. However, they do not work in isolation; they have overlapping sectors of fire. This means that even in the same muscle fiber, aerobic and anaerobic systems are working together to fuel movement. Moreover, due to the above explanation of fiber type recruitment, the energy systems will also exchange duties as the overall primary energy pathway as you speed or slow the acceleration of your movement. With proper training, you can increase the strength of your slower fibers and increase the aerobic capacity of your faster fibers, and this is the core of strength & endurance training.
Oddly, aging and a sedentary lifestyle tend to harvest the faster fibers and reduce the aerobic qualities of the slower fibers, leaving the sedentary older adult with weak, sugar-burning muscle tissue. This explains a lot of the physical performance problems observed that are associated with the aging population. To some extent, strength training will also increase the endurance of the faster fibers and endurance training will increase the strength of the slower fibers. There is no reason, at this point, to prescribe one or the other interventions: Either one will stimulate positive adaptations in both strength and endurance. Especially with the aging and sedentary population, a physical training protocol that both improves the strength and endurance of all types of muscle fibers is warranted.
The A+A protocol falls under a larger umbrella of “anti-glycolytic” training that targets exactly the above idea, resulting in improvements in both strength and endurance while reducing the stress and biologic impact of the training sessions. This leaves you fresh to deal with the other tasks in life without having to drag you’re a** through the rest of your day.
Supporting Literature:
- Baker, McCormick, & Robergs, 2010.
- Keyser, 2010.
- Rogatzki, et al., 2015.
David says:
Wow. An amazing level of detail. Let’s say I want to try out an A&A training protocol. What would I do? Thanks in advance!
Al Ciampa says:
Use the contact page on this website to send me a message. -Al
Zuzane says:
Hi Mr.Ciampa Iam train for long long time with Kettlbells can you give some simple combination of exercises what you do ?
Or just give some simple scheme?
Are you think that A+A with snatches is enough or can I add some push ups or TGU or Loaded Carries?
Thank you
Appreciate what you do for out sport
Best
Zuzan
Al Ciampa says:
Hi Zuzan,
Thanks for your question. A+A is more about the volume of power work with sufficient recovery during the session. Exercise choice is a matter of what works best—like the KN snatch.
All of those supplementary choices are fine to incorporate.
Jim Sullivan says:
Excellent resource. Thank you.